REGULATION OF OBESITY AND INSULIN RESISTANCE BY NITRIC OXIDE
This is a very comprehensive journal article on Nitric Oxide’s impact on IR & obesity!
It makes a very handy reference guide. The only part I find objectionable is the author’s take on obesity being an energy imbalance issue where it is probably better described as an energy storage and access issue.
It does use slightly different nomenclature for the various isoforms.
NOS1 = nNOS (neuronal)
NOS2 - iNOS (inducible associated with inflammation response)
NOS3 = eNOS (endothelial)
Interesting that eNOS (and presumably dietary nitrate derived NO) suppresses GNG! That could be a handy tool in the keto toolbox!
6. Summary
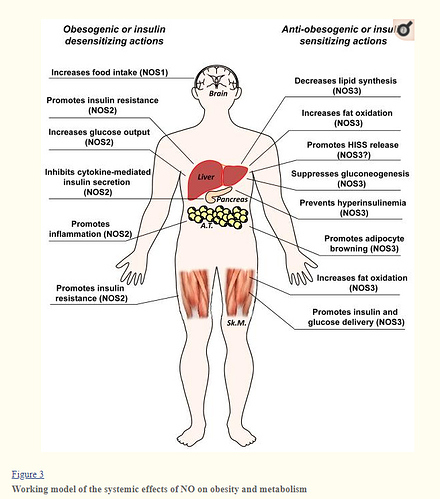
Integration of results from these studies helps to form a model illustrating the role of NO in regulating obesity and insulin resistance (Figure 3). NO derived from NOS3 appears to have both anti-obesogenic and insulin-sensitizing properties. Its anti-obesogenic role stems from its ability to increase fat oxidation in peripheral tissues such as skeletal muscle, liver, and adipose tissue. As mentioned above, there is evidence that it also decreases lipid synthesis in liver. The impact of NOS3 on glucose metabolism and insulin sensitivity is underpinned by its capacity to increase transport of insulin and glucose to key peripheral tissues such as skeletal muscle and to regulate gluconeogenesis. Additionally, there may be implications for NOS3-mediated HISS release, which appears to enhance the vasodilatory properties of insulin. That NOS3 prevents hyperinsulinemia in two independent genetic gain-of-function studies [122, 337] further suggests that it could impact glucose metabolism by modulating insulin release. Other isoforms of NOS appear to promote deleterious changes in metabolism. In the brain, evidence suggests that NOS1-derived NO promotes hyperphagia. The NOS2 isoform promotes insulin resistance in both liver and skeletal muscle and is critical in inflammatory responses in multiple tissues, most notably, the adipose organ. Counter to NOS3, NOS2 appears to promote gluconeogenesis, and NOS2 has remarkable effects on cytokine-mediated insulin secretion. From these studies, it is apparent that NO is one of the most critical features regulating metabolism, body composition, and insulin sensitivity. Harnessing its beneficial metabolic actions is an exciting prospect for combatting metabolic disease.



 It was hard too for me cause I lost lbs fast and then nothing. total stall out for a very long time til one day, boom, I started losing again but at a crawl pace too but I am cool with that slower pace cause zc is my life long eating plan so I accept it. Super congrats on your big loss, that is quite an accomplisment!! But going back into a keto plan if ya love it and works well for you is great!
It was hard too for me cause I lost lbs fast and then nothing. total stall out for a very long time til one day, boom, I started losing again but at a crawl pace too but I am cool with that slower pace cause zc is my life long eating plan so I accept it. Super congrats on your big loss, that is quite an accomplisment!! But going back into a keto plan if ya love it and works well for you is great!