In my opinion you could be in ketosis and you might randomly burn body fat and you could be eating a high carb diet and you might randomly burn body fat. Either way you could store fat then burn it over and over again in the fatty acid turnover pool so it would appear your not doing anything and creep onto the side of gaining?
Insulin might store glucose as body fat or help burn it before the excess glucose is stored!
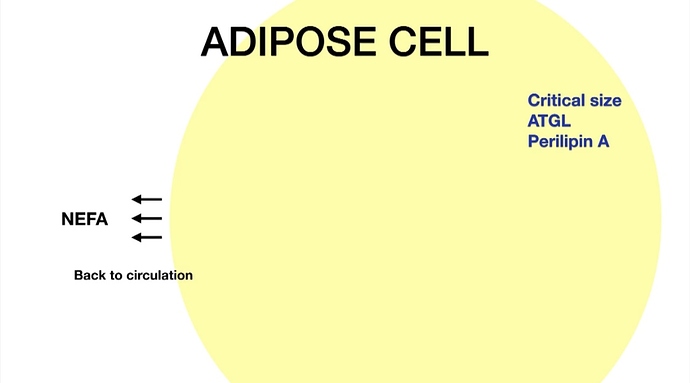
Too much reliance on information derived about insulin and glucagon is the problem because insulin and glucagon is only co-factor in lipolysis.
It almost seems like the goal is getting the body to produce more adrenaline (HPA -axis activation), but is that what actually burns body fat?
If you can scare the body into survival mode by starving it or some how stimulate the components that makeup adrenaline?
I’m still examining the research on this (truly mind boggling stuff).
Footnotes:
[1] Vascular and metapbolic effects of adrenaline in adipose tissue in type 2 diabetes: “… Capillaries in the adipose tissue are recruited by adrenaline in non-T2DM subjects; however, this response is impaired in overweight T2DM subjects. NEFA, released in adipose tissue during adrenaline stimulation, is insufficiently re-esterified in situ in overweight T2DM subjects, probably owing to increased ATBF after adrenaline infusion and inability to increase adipose tissue glucose uptake.
[3] Effect of pH on epinephrine-induced lipolysis in isolated fat cells
[4] Lipolysis in adipocytes is under tight hormonal control. Catecholamines interacting with β-adrenergic receptors stimulate lipolysis and insulin counters this to suppress lipolysis (Holm et al. 2000). NAFLD patients exhibit an inability for insulin to suppress peripheral lipolysis, contributing to increased FFA in the circulation (Sanyal et al. 2001). In vivo stable isotope studies in NAFLD patients also suggest that inappropriate lipolysis in adipose tissue contributes to the triglycerideaccumulated within the liver (Donnelly et al. 2005). While no data are available from humans with ALD, in animal models chronic ethanol feeding results in a dysregulation of lipolysis in adipose tissue. Chronic ethanol impairs the ability of insulin to suppress adipose lipolysis, resulting in a decrease in the rate of glycerol appearance into the circulation (Kang et al. 2007a). This insulin resistance is observed both in adipocytes isolated from ethanol-fed rats and in vivo under the controlled conditions of a hyperinsulinemic-euglycemic clamp (Kang et al. 2007a). In studies using 2H2O as a tracer, the rate of triglyceride turnover in epididymal adipose tissue was increased after ethanol feeding (Kang et al. 2007a). Chronic ethanol feeding increased triglyceride degradation by 2.3-fold compared to pair-fed rats, but had no effect on triglyceride synthesis (Kang et al. 2007a). Taken together, these data indicate that a profound dysregulation of lipolysis in adipose tissue likely contributes to the progression of hepatic steatosis in both NAFLD and ALD.
View chapter …More






 Eating doesn’t necessarily stop ketosis, no? If one eats sufficient carbs to stop one from using fat for energy, then sure, but if one is eating ketogenically and using fat for energy, why would ketosis stop?
Eating doesn’t necessarily stop ketosis, no? If one eats sufficient carbs to stop one from using fat for energy, then sure, but if one is eating ketogenically and using fat for energy, why would ketosis stop?