Body fat loss / metabolism and heat. @carl @richard
Just want to be clear on the Conservation of Mass.
When a person loses body fat the carbon, oxygen and hydrogen atoms change from fat molecules to carbon dioxide and water*. At least according to science communicator Ruben Meerman.
The fat molecules do not turn into heat, per se. Their complex bonds cleave to create the energy, but the atoms just become part of smaller molecules.
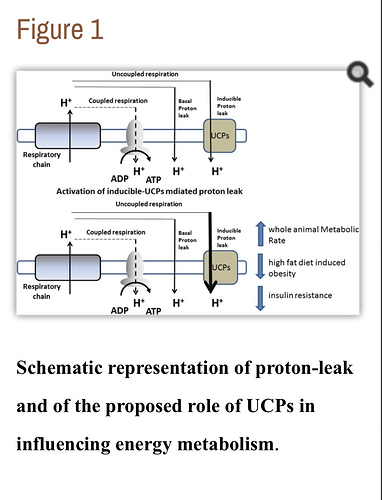
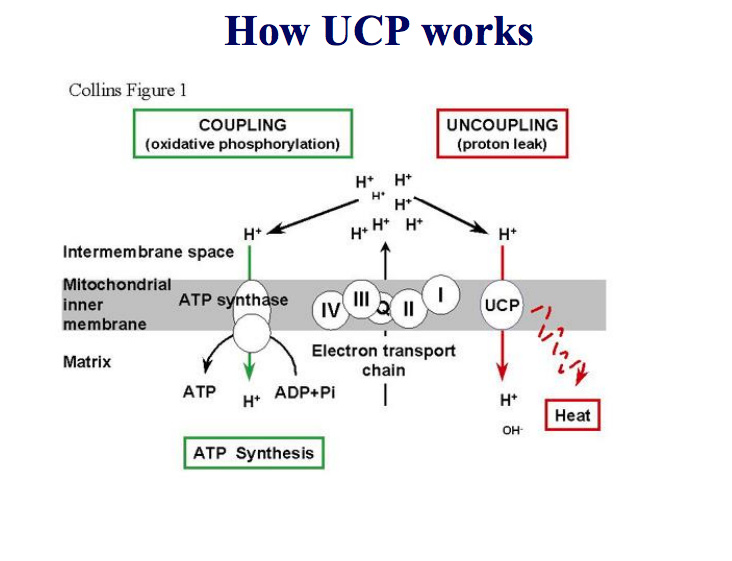
The uncoupled (corrected from ‘decoupled’) mitochondria can still work to generate more cellular ATP energy and generate more carbon dioxide and water to be excreted.
*But I reckon ketone molecules with their higher molecular weight, than carbon dioxide and water, being spilled out of the body as acetone (breath) and via ketones in the urine would be part of the metabolic advantage in nutritional ketosis for weight loss in type 2 diabetes treatment.
In the discussion it sounded like, to me anyway, the ketones were being burned for heat energy, which could be misinterpreted literally as fat turning into heat.
Where there’s fire, one should also consider the smoke.