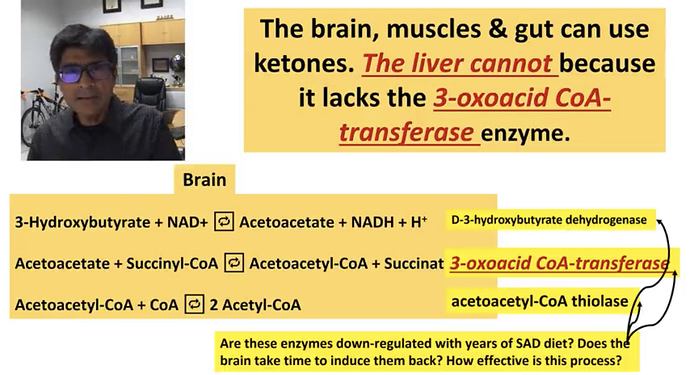
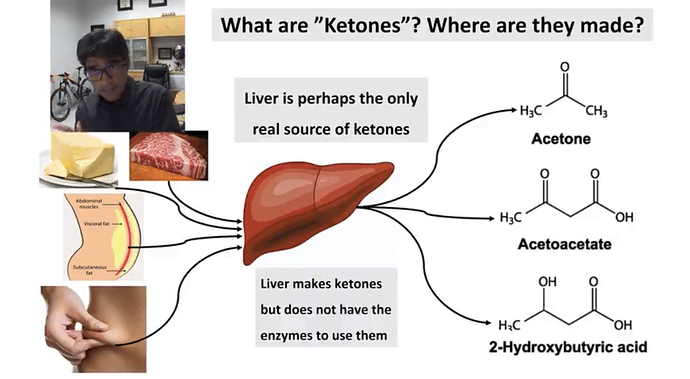
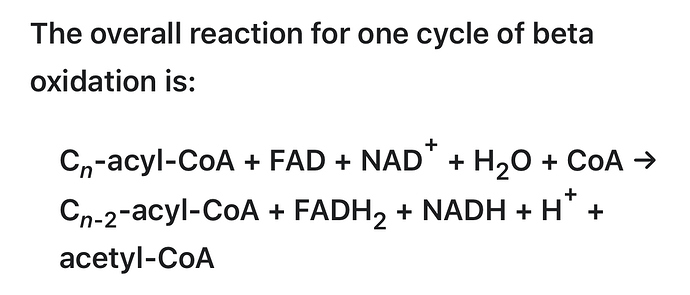I’m resurrecting this topic to reference the following link. Unlike Chris Masterjohn ref’d above by @brownfat, I do not see this genetic variation as being any indication/evidence that our ancestors were not nor intended by nature to be in continuous, long term, life-long ketosis. Masterjohn is basically buying into the argument that ketosis is an unusual, emergency metabolic state to tide us over until we can start eating carbs again. I think that argument fails because there is little/no evidence that prior to the agricultural revolution carbohydrates were anything more than a minor supplement to the primarily meat/fat diet of our hominid ancestors for hundreds of thousands if not several million years. See this.
I find the argument implausible that a subgroup of humans who lived for tens of thousands if not hundreds of thousands years in an environment in which carbohydrates were totally absent developed a genetic mutation that made them dependent upon consuming carbohydrates to survive. They would have gone extinct within a year of developing such a thing. Yet, instead, they thrived for thousands of generations. I’m willing to accept that we probably don’t yet know exactly what went on for all those thousands of years and generations of Inuit. But what I’m very sure did not happen was that this genetic variation impeded their ability to metabolize fats in any significant manner and probably enhanced that ability in some way.
CPT1A imports long-chain fatty acids into mitochondria for use in fatty-acid oxidation. This helps to maintain energy homeostasis and normoglycemia when carbohydrate intake is low.27 The extent to which the c.1436C>T mutation contributes to disorders associated with CPT1 deficiency, such as hypoketotic hypoglycemia and sudden infant death syndrome, is still unclear. The derived allele has been reported as being deleterious in both the homozygous and the heterozygous state. Yet, its phenotypic effect might depend upon many environmental factors, e.g., feeding history, infection, and climate.13, 27 It is known that the mutation decreases fatty-acid oxidation and ketogenesis, explaining its role in hypoketotic hypoglycemia.13, 28 However, there is also evidence that the mutation decreases the inhibitory effect of malonyl-CoA on fatty-acid β-oxidation in mitochondria, thereby partially compensating for the drop in ketogenesis associated with reduced CPT1A activity.13, 28 A study on Alaskan Yup’iks also suggests that the c.1436C>T mutation might exert a cardioprotective role through its association with elevated levels of high-density lipoprotein cholesterol and reduced adiposity.28 Moreover, the large amounts of n-3 polyenoic fatty acids in the traditional diet of these aboriginal peoples are known to increase the activity of CPT1A.13, 28 In this context, the CPT1A-activity decrease due to the c.1436C>T mutation could be protective against overproduction of ketone bodies.13 These important metabolic effects of CPT1A provide the basis of our hypothesis that the c.1436C>T mutation might have conferred a metabolic advantage for the Northeast Siberian populations in dealing with their traditional high-fat diet. The deleterious effect of the mutation might be explained by a change from the traditional diet to a more carbohydrate-based one or by recent cultural shifts and environmental stressors such as fasting and pathogens.
In conclusion, CPT1A c.1436C>T joins the short list of known human variants where ill health in present-day populations is a likely consequence of the same variant’s being selectively advantageous in the past. Compared with the sickle cell allele rs334 (associated with sickle cell disease [MIM 603903] and malaria resistance [MIM 611162])31 or rs73885319, rs60910145, and rs71785313 in apolipoprotein L-I ( APOL1 [MIM 603743], associated with kidney disease [FSGS4 (MIM 612551)] and sleeping-sickness resistance32), the c.1436C>T allele shares the property of altering a protein sequence. However, unlike the sickle cell allele, it does not represent an example of heterozygous advantage but instead provides an advantageous or disadvantageous effect dependent on the environment. In this way, it extends the range of selective forces contributing to current ill health beyond infectious diseases. It illustrates the medical relevance of an evolutionary understanding of our past and suggests that evolutionary impacts on health might be more prevalent than currently appreciated.